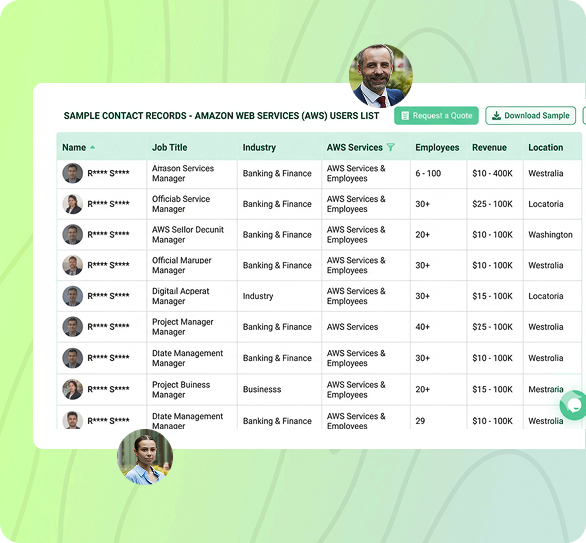
Oracle Health Sciences Market Position by Category
| Category | Position | Notes |
|---|---|---|
| Pharma EDC (Top 20 Sponsors) | Co-leader with Veeva and Medidata | InForm and Oracle Clinical have decades of installed base in big pharma |
| Pharmacovigilance & Drug Safety | #1 globally | Argus Safety is the de-facto standard at most top 50 pharma companies |
| CTMS for Mid-Market Pharma | Top 3 | Siebel CTMS competes with Veeva Vault CTMS and Medidata CTMS |
| Clinical Signal Detection | Market leader | Empirica Signal is widely used by regulators and major pharma |
| Real-World Evidence Platforms | Top 5 | Oracle Health Sciences Cloud expanding aggressively here |
*Disclaimer: Position assessments reflect installed base presence in our database, not exhaustive market share studies. Information is subject to change as the life sciences software market evolves rapidly.